

What is AMNIOEFFECT?
AMNIOEFFECT® is a placental allograft comprised of human amnion, intermediate layer, and chorion that provides a protective barrier on surgical wounds and exposed tissues. It is available in a wide variety of sizes to meet surgeons’ clinical needs.
AMNIOEFFFECT Product Overview
Protective Environment
AMNIOEFFECT provides a protective barrier on wounds and thereby supports development of granulation tissue and the healing process.
Product Advantages
- Human-derived tissue
- The PURION® process preserves Extracellular Matrix (ECM) components including 300+ regulatory proteins1
- Thickness of allograft allows for suturing to keep the graft in place and for placement in deeper surgical wounds*
- Shelf stable^
- Available in sheet configurations up to 180 cm2 to address a variety of surgical wounds
- Compatible with Negative Pressure Wound Therapy (NPWT) and Hyperbaric Oxygen Therapy (HBOT)
^ See Instructions for Use
Clinical Use Examples in Surgeons’ Practices
- Amputations
- Complex incision management
- Dehiscence repair
- Tendon & ligament repair
- Exposed bone or hardware
- Flaps
- Laminectomies
- Minimally Invasive Surgery (MIS)
- Hysterectomy
- Endometriosis surgery
- Pilonidal cyst removal
*Not intended for use as a load bearing tissue.
Making a Difference
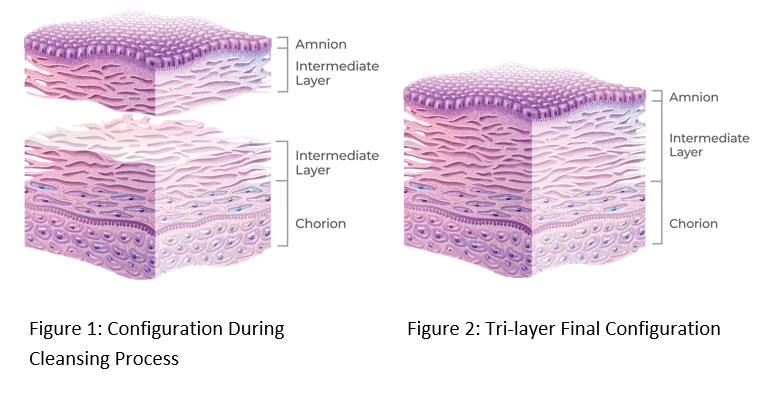
AMNIOEFFECT Processing & Details
The PURION process has been optimized for retention of the Intermediate Layer (IL). Separation of the amnion and chorion, with attached IL (Figure 1), ensures thorough cleansing of each layer prior to lamination into the tri-layer final configuration (Figure 2).
Resources
AMNIOEFFECT FAQ: Meshing Product
MIMEDX Advantage
We improve people’s health and lives through innovation that delivers solutions for patients and caregivers. With a wide variety of allograft configurations, clinicians can choose the product that best meets their treatment objectives.
Healthcare Professionals
MIMEDX offers unique value to clinicians across sites of care. With a broad support system, we offer services to support needs relating to reimbursement and coverage, product access and cost containment, education, patient information, and clinical applications.
Our Products
MIMEDX offers a broad portfolio of advanced wound and surgical products, giving clinicians the flexibility to choose the right product for every patient and clinical setting.
References
- Moreno S, Massee M, Campbell S, Bara H, Koob TJ, Harper JR. PURION processed human amnion chorion membrane allografts retain material and biological properties supportive of soft tissue repair. J Biomater Appl. 2024;39(1):24-39.

