
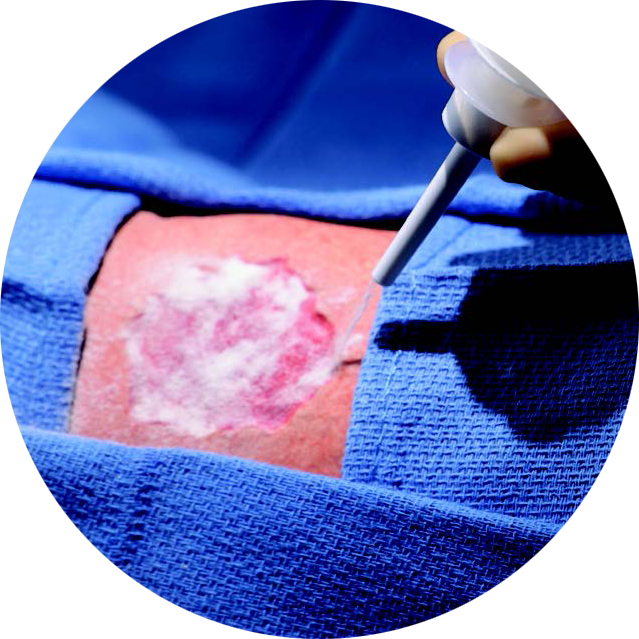
Hydrelix™ Collagen Matrix is a Type I collagen powder made from hydrolyzed modified bovine collagen for the management of wounds.
Hydrelix Collagen Matrix, formulated from hydrolyzed and modified collagens in powdered form, provides immediate, high-solubility peptides to support a moist healing environment.
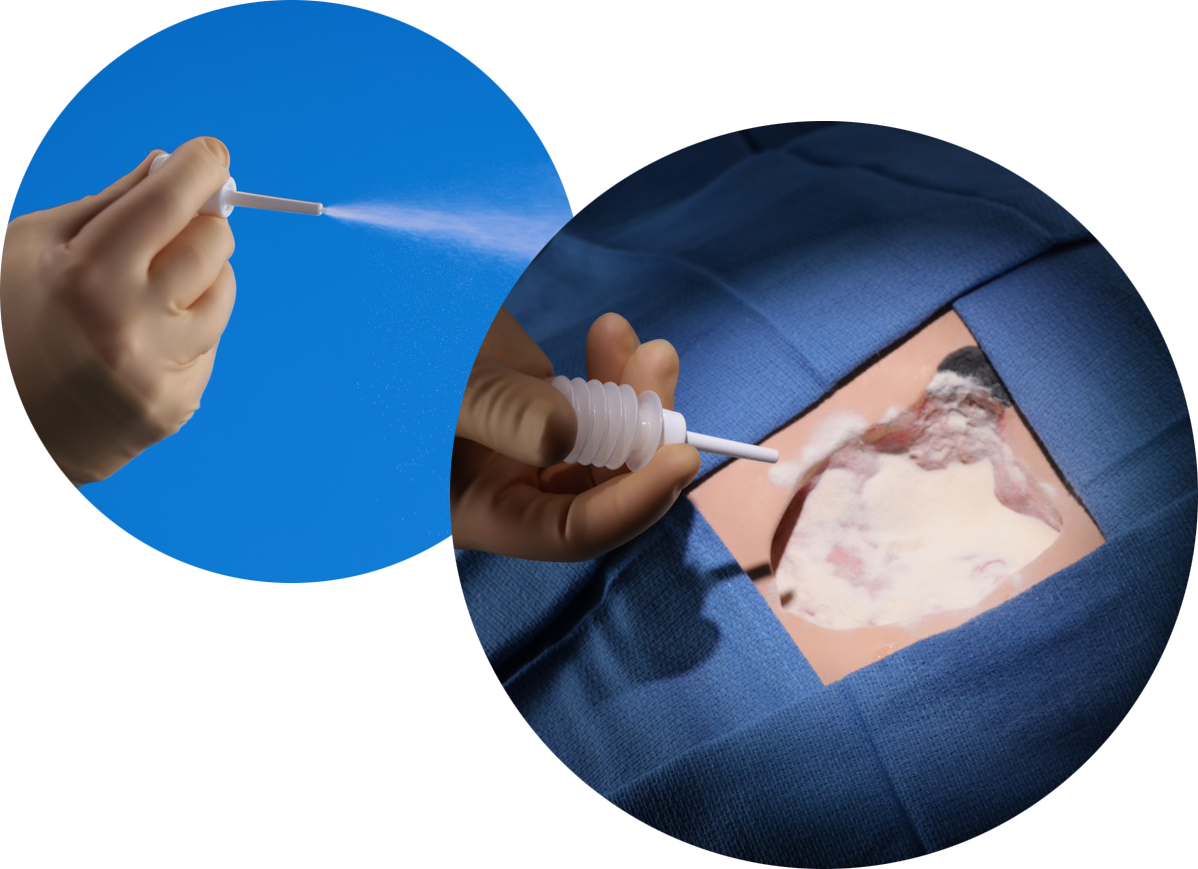
Key Benefits and Applications
- Composed of hydrolyzed, modified collagens formulated to dissolve in wound exudate
- Forms a gel that creates a moist environment conducive to healing
- Enzymatically degraded by wound exudate and provides high-solubility peptides
- Suitable for surgical wounds across diverse specialties
- Maintains a moist environment conducive to tissue regeneration
- Advanced accordion dispenser ensures precision placement of collagen and reduces the risk of human contamination
Hydrelix Collagen Matrix is indicated for the management of wounds including:
- Full and partial thickness wounds
- Pressure ulcers (stages I-IV)
- Venous stasis ulcers
- Diabetic ulcers
- Partial thickness burns
- Traumatic wounds healing by secondary intention
- Acute wounds
- Abrasions
- Donor sites and other surface wounds
Hydrelix
Collagen Matrix
| GTIN | Product # | Description | Size |
| 0745713000014 | HDX-001 | Hydrelix Collagen Matrix | 1 g |
| 0745713000045 | HDX-005 | Hydrelix Collagen Matrix | 5 g |
Indications For Use: Hydrelix is indicated for prolonged use (24hrs to 30 days) in the management of wounds such as partial and full thickness wounds, pressure ulcers (stages I-IV), venous stasis ulcers, diabetic ulcers, partial thickness burns, acute wounds, abrasions, traumatic wounds healing by secondary intention, and donor sites and other surface wounds.
Healthcare professionals must use their own clinical judgment in evaluating appropriate treatment options for a particular patient. Treatment of a specific patient should be based on individual needs and the medical care deemed necessary by the patient’s treating physician and institutional protocols. Always refer to the package insert, product label, and/or instructions for use before using any MIMEDX product. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your MIMEDX representative if you have questions about MIMEDX products.
Hydrelix is manufactured by Southwest Technologies, Inc. MIMEDX is the exclusive distributor of Hydrelix. MIMEDX is a trademark owned by MIMEDX Group, Inc. All other trademarks are owned by their respective owners.








