Allograft Protective Barriers in Spine and Neurosurgery
Spine revisions and cranioplasties can be complicated by difficulty of re-accessing the tissue planes resulting in potential adverse outcomes postoperatively.1 Additionally, postop wound-related complications are the most common cause of readmission in spine surgery.2
Peer-reviewed RCTs and systematic studies have reported favorable results in patients undergoing cranial and spine surgeries with human amniotic membrane allografts being used as a protective barrier on acute wounds (e.g., exposed dura) and helps separate the planes of tissue.3
Single-Center Retrospective Outcomes With AMNIOFIX® as a Protective Barrier
- In spine acute wound incision case studies (n=4), AMNIOFIX recipients showed complete wound resolution at 6 weeks postop.4
RCT Outcomes With DHACM (Dehydrated Human Amnion Chorion Membrane) as a Protective Barrier
- DHACM recipients have shown high closure rates in challenging wounds such as DFUs and VLUs with statistical significance vs. SOC.5-14
Surgeon Testimonials
“I have noticed a significant reduction in difficulty in reoperations when using AMNIOFIX amnion/chorion allografts as a protective barrier in a variety of spine procedures.”
Besty Grunch, MD | Neurosurgeon | Gainesville, GA
“Patients that have undergone craniectomy have greatly benefited in receiving AMNIOFIX as a protective barrier because there has been a good tissue plane that can be easily developed without much effort during cranioplasty.”
Bryan Lee, MD | Neurosurgeon | Pheonix, AZ
Examples of AMNIOFIX Used as a Protective Barrier
Laminectomy & Fusion
Microdiscectomy
Craniectomy
Product Advantages: AMNIOFIX and AMNIOEFFECT®
- Provides a protective barrier on wounds and thereby supports the healing process
- Terminally sterilized for additional level of safety
- PURION® process preserves Extracellular Matrix (ECM) components, including 300+ regulatory proteins15,16
- Easy to apply
- Shelf-stable*
- Up to 5-year shelf life
- Does not expand upon hydration17
Examples of AMNIOFIX Used as a Protective Barrier in Surgeons’ Practices
SPINE
- Laminectomies
- Fusions
- Revisions
- Microdiscectomies
-
Endoscopic spine procedures
-
Spine acute wound incisions
*See Instructions for Use
NEUROSURGERY
- Craniectomies
- Craniotomies
- Cranioplasties
Case Studies and Clinical Evidence:
How Physicians Use Our Products
spine case study
Posterior Lateral Fusion With Decompression and Application of AMNIOFIX AS A PROTECTIVE BARRIER18
Patient Background
69-year-old male treated for neurogenic claudication for 2 years with conservative measures (epidural steroid shots) and started to develop left ankle dorsiflexion weakness.
Surgical Intervention
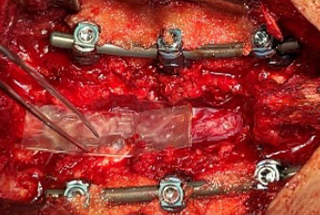
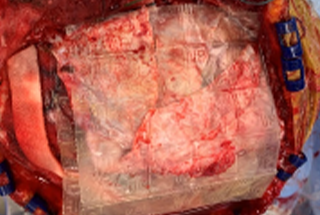
- Patient underwent a lateral interbody fusion at L3-L4 and L4-L5 and a laminectomy L3-L5 to achieve a decompression.
- Prior to closure, a 4 cm x 6 cm AMNIOFIX sheet was cut lengthwise into two pieces.
- The first 2 cm x 6 cm AMNIOFIX sheet was applied as a protective barrier to the exposed dura and tucked underneath the lamina and lateral recesses.
- The second 2 cm x 6 cm AMNIOFIX sheet was applied as a protective barrier on the cranial aspect of the exposed dura. The allograft was then tucked into the lateral gutters and underneath the cephalad lamina.
Goal
AMNIOFIX utilized to provide a protective barrier and thereby supported the healing process.
Conclusion
- The dura was decompressed and no postop complications were reported. The surgeon and patient were pleased with the outcome.
- The surgeon anticipated that this patient will likely have adjacent level disease to the treated area that would be symptomatic in 5 to 10 years and may require an adjacent level decompression.
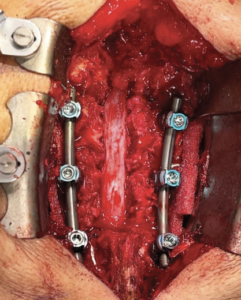
Lateral interbody fusion L3-L4 and L4-L5 and laminectomy L3-L5. |
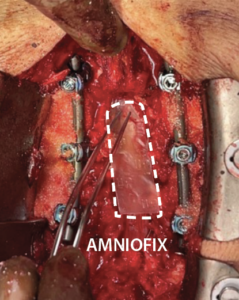
4 cm x 6 cm AMNIOFIX sheet was cut lengthwise into two pieces. The first 2 cm x 6 cm AMNIOFIX sheet was applied as a protective barrier to the exposed dura and tucked underneath the lamina and lateral recesses. |
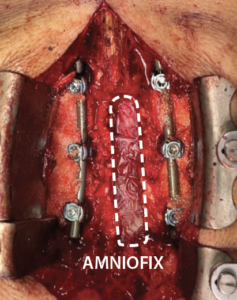
The second 2 cm x 6 cm AMNIOFIX sheet was applied as a protective barrier on the cranial aspect of the exposed dura. Allograft was tucked into the lateral gutters and underneath the cephlad lamina. |
CRANIOPLASTY Case Example Videos
AMNIOFIX has been utilized as a protective barrier on wounds in decompressive craniectomy procedures.
The following cranioplasty videos feature patients who previously received AMNIOFIX during their initial decompressive craniectomy surgeries.
Each cranioplasty procedure shown in the videos is completed in under three minutes.
|
Bilateral Frontal Cranioplasty With Previous AMNIOFIX Placental Allograft Protective Barrier Recipient
|
Cranioplasty With Previous AMNIOFIX Placental Allograft Recipient
|
Case(s) based on clinician experience. For educational purposes only. Not meant to direct clinical decision making. See Instructions for Use for complete information including tissue use.
AMNIOFIX is not intended to repair dura mater and not intended as a dura substitute.
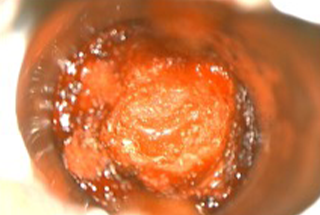
Spine Acute Wound Incision Closure in Comorbid Patient
L5-S1 Posterior Fusion: Acute Wound Incision Closure in Comorbid Patient5
Clinical History: 71-year-old female with a mobile spondylolisthesis and a large synovial cyst.
Comorbidities: Obesity (BMI 31) and osteopenia.
Surgical Intervention: L5-S1 posterior pedicle screws and fusion with open decompression for removal of right synovial cyst. AMNIOFIX provided a protective barrier on wounds and thereby supported the healing process. Primary closure with Vicryl® absorbable suture and skin adhesive applied over sutured incision.
Results
- Incision was intact and closed at 6 weeks postop.
- Patient and healthcare team were very satisfied with the outcome of the closed incision.
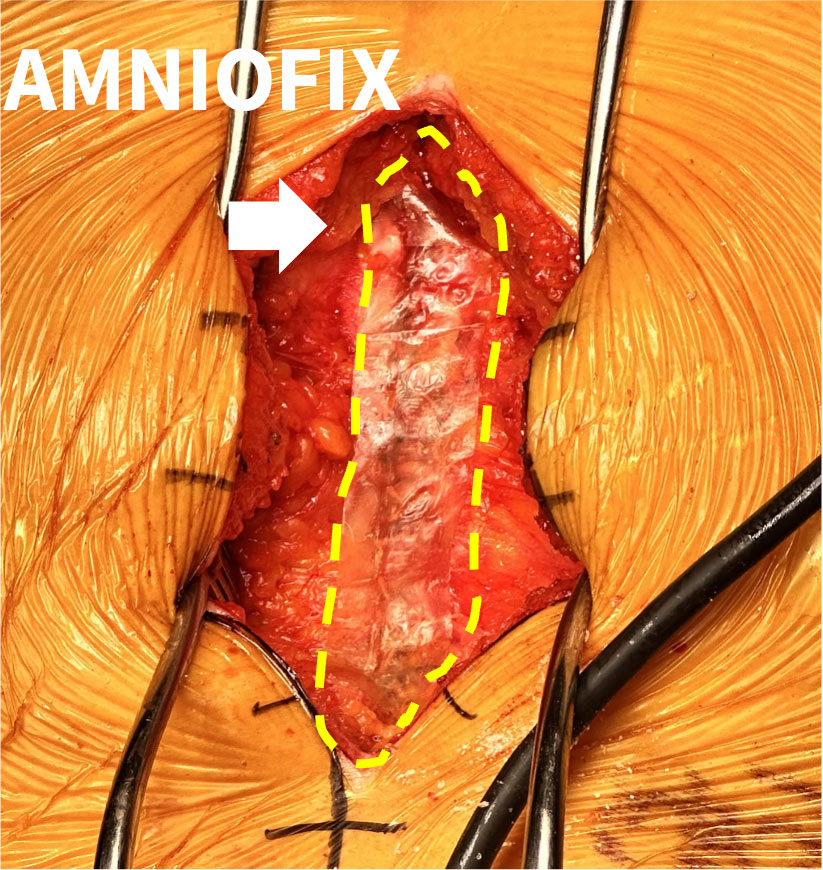
Intraop placement of AMNIOFIX |
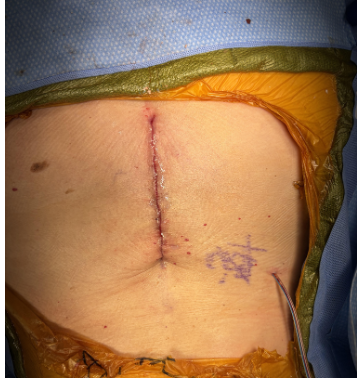
Day 0: Postop |
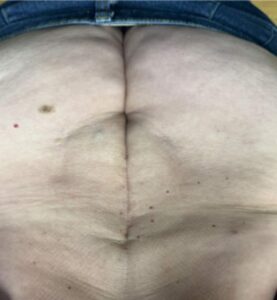
6 Weeks: Postop |
Note: BMI = Body Mass Index
Surgical Product Portfolio: Advanced Placental Allografts
MIMEDX offers a portfolio of advanced placental allografts in the surgical setting.
- AMNIOFIX
- AMNIOCORD®
- AMNIOEFFECT
- AMNIOBURN®
- AXIOFILL®
References
- Lee B. MIMEDX interview with Bryan Lee, MD. October 4, 2023.
- Bernatz JT, et al. Neurosurgical Focus FOC. 2015;39(4):E7.
- Moore ML, et al. Int J Spine Surg. 2023;17(1):32-42.
- Betsy Grunch, MD, Neurosurgeon, Gainesville, GA.
- Zelen CM, et al. Int Wound J. 2013;10(5):502-507.
- Zelen CM. J Wound Care. 2013;22(7):347-351.
- Zelen CM, et al. Wound Medicine.2014;4:1-4.
- Zelen CM, et al. Int Wound J. 2014;11(2):122-128.
- Zelen CM, et al. Int Wound J. 2015;12(6):724-732.
- Zelen CM, et al. Int Wound J. 2016;13(2):272-282.
- Tettelbach W, et al. Int Wound J. 2019;16(1):19-29.
- Serena TE, et al. Wound Repair Regen. 2014;22(6):688-693.
- Bianchi C, et al. Int Wound J. 2018;15(1):114-122.
- Bianchi C, et al. Int Wound J. 2019;16(3):761-767.
- Moreno S, et al. J Biomater Appl. 2024;39(1):24-39.
- MIMEDX Internal Report. MM-RD-00086.
- MIMEDX Internal Report. MM-RD-00193.
- Jeff Phelps, MD, Spine Surgeon, Dallas / Ft. Worth, TX.