Applications in Gynecologic Surgery
Advanced Placental Allografts When Patients are in Need
There are many risk-factors to healing such as patient co-morbidities and complex defects.1 MIMEDX offers an array of advanced placental allografts that may be used as a protective barrier in a variety of surgical applications to provide valuable benefits in patient care.
AMNIOFIX® Application
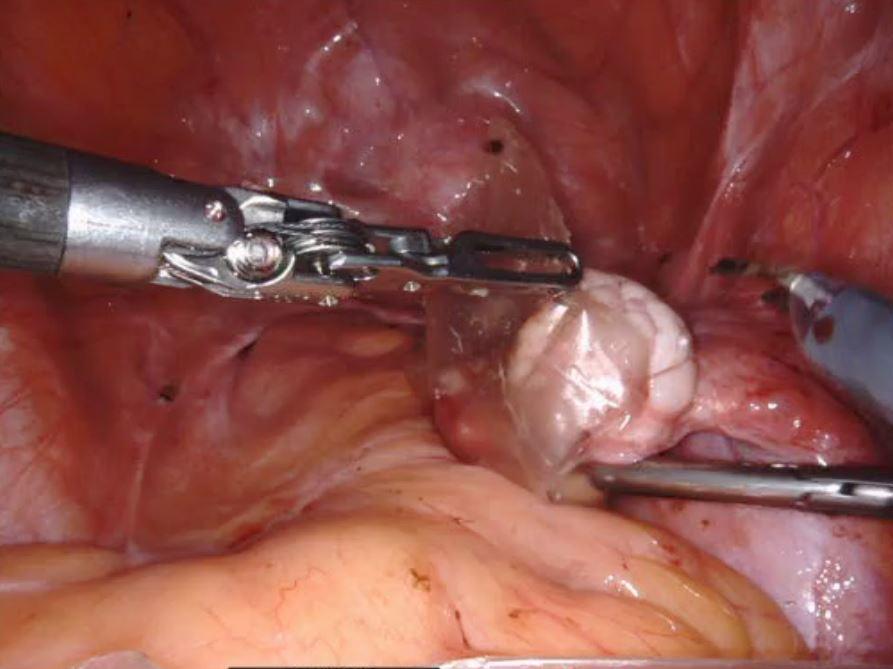
Product Advantages: AMNIOEFFECT®, AMNIOFIX, and AMNIOCORD®
- Provide a protective barrier on wounds and thereby supports development of granulation tissue and the healing process
- Terminally sterilized for additional level of safety
- AMNIOFIX & AMNOEFFECT: PURION® process preserves Extracellular Matrix (ECM) components, including 300+ regulatory proteins2,3
- AMNIOCORD: PURION process preserves ECM components, including 250+ regulatory proteins4
Clinical Use Examples:
- Hysterectomies
- Myomectomies
- Endometriosis
- Debridement/ dehiscence
- Complex incision management
Case Studies:
How Physicians Use Our Products
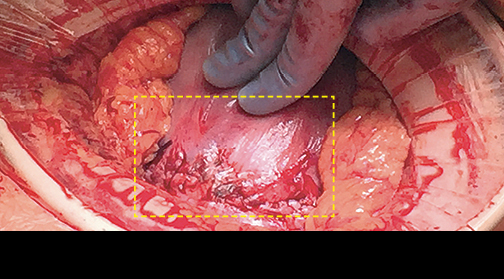
Case study 1: Endometriosis
Endometriosis Resection with AMNIOFIX5
Peer Reviewed Publication Overview
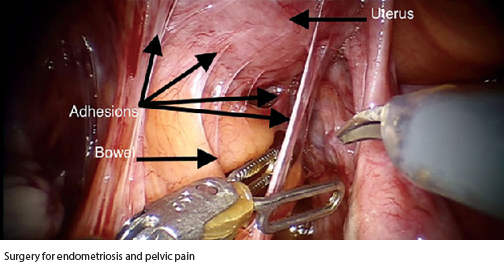
- Retrospective case series in 16 consecutive patients with surgery for endometriosis and pelvic pain.
- 31.2% of patients were obese and mean number of prior surgeries was 4.1 +/- 3.2.
- All 16 patients had significant abdominal adhesions at the time of surgery.
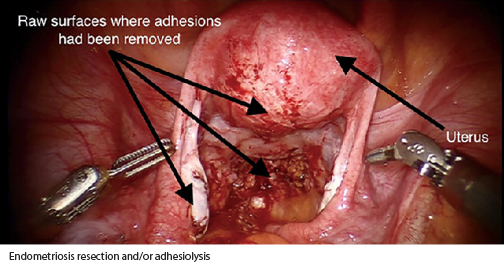
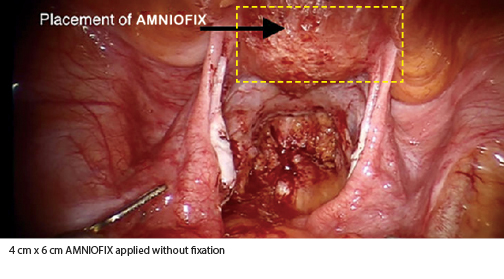
- One or more 4 cm x 6 cm AMNIOFIX grafts were placed without fixation after endometriosis resection and/or adhesiolysis.
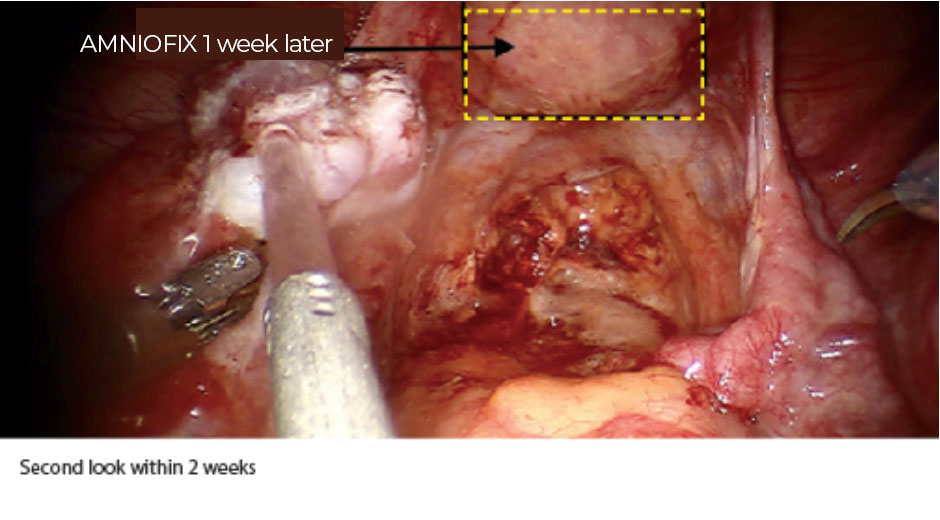
- 2nd surgical procedure within 2 weeks to inspect for reformation of adhesions and lyse any additional endometriosis lesions or adhesions found.
Observed Outcomes
- At the second surgery, approximately 93% of patients previously treated with AMNIOFIX demonstrated good tissue quality and clearly separated tissue planes (14 of 15 patients; one of the 16 patients canceled).
- In one AMNIOFIX recipient, tissue quality was reduced and some tissue planes remained attached. However, these planes separated more easily over the AMNIOFIX-treated area than in adjacent untreated tissue.
- Only one AMNIOFIX allograft shifted from its original placement. Attached tissue planes were noted near, but not directly on, the displaced allograft.
- No adverse events related to AMNIOFIX use were reported.
 |
 |
 |
 |
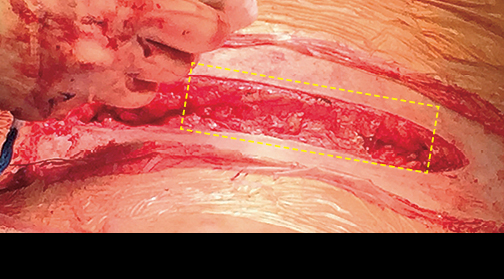
Case study 2: Caesarean
Caesarean Section with AMNIOFIX6
Clinical History
31-year-old G4, P2012 with a BMI of 46 scheduled for her 3rd full-term C-section and bilateral salpingectomy.
Challenge
Adhesions were expected after two prior full-term C-section deliveries, and there were concerns for postoperative wound complications due to the patient’s obesity and large pannus.
Surgical Intervention
Dense adhesions were present while gaining access to the uterus as well as between the bladder and the uterus. Adhesiolysis left a defect in the uterus, and bilateral salpingectomy was performed. AMNIOFIX was placed on each tubal excision site and the uterus defect closure. (NOTE: Normally this surgeon would have preferred to place AMNIOFIX on the entire uterine suture line, especially if a future C-section was expected.)
After fascial closure, a 2 cm x 12 cm AMNIOFIX graft was also placed in the incision site and then the skin was closed.
Follow-Up
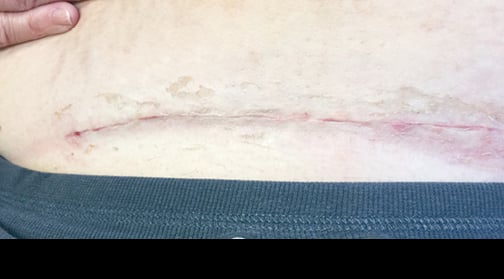
At 10-day follow-up, the incision was fully closed without signs of infection. This was impressive considering the large pannus fully covering the incision site. The patient also indicated that she had a faster recovery compared to her prior deliveries.
|
AMNIOFIX placed in subcutaneous space after fascial closure
|
AMNIOFIX placed on uterus detect closure |
Incision 10 days postop
|
Case study 3: Keloid Scar
Keloid Scar Revision with EPIFIX®7
Clinical History
Patient presented with keloid scar after Caesarean section procedure.
Treatment
EPIFIX is a dehydrated human amnion/chorion membrane allograft. The product provides a protective barrier on wounds and thereby supports development of granulation tissue and the healing process. The PURION process preserves ECM components, including 300+ regulatory proteins.2,3
Follow-Up
At year follow up, the one-third portion of the scar that was treated was greatly reduced in height and in color. Subsequent revision surgery managed the remaining two-thirds of the keloid scar with EPIFIX used to cover and protect the area.
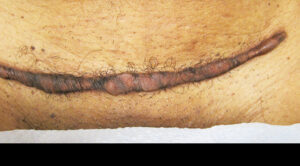
Operative Presentation |
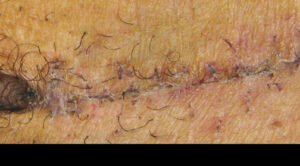 Revision using EPIFIX on 1/3 portion of original scar Revision using EPIFIX on 1/3 portion of original scar |
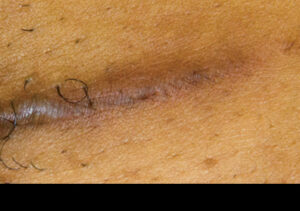 Scar one year post surgical treatment with EPIFIX overlay Scar one year post surgical treatment with EPIFIX overlay |
Resources
Surgical Product Portfolio: Advanced Placental Allografts
- AMNIOFIX
- AMNIOCORD
- AMNIOEFFECT
- AMNIOBURN®
- AXIOFILL®
References
- Atkin L, Bućko Z, Conde Montero E, et al. Implementing TIMERS: the race against hard-to-heal wounds. J Wound Care. 2019;23(Sup3a):S1-S50.
- Moreno S, Massee M, Campbell S, Bara H, Koob TJ, Harper JR. PURION® processed human amnion chorion membrane allografts retain material and biological properties supportive of soft tissue repair. J Biomater Appl. 2024;39(1):24-39.
- MIMEDX Internal Report. MM-RD-00086, Proteome Characterization of PURION Processed Dehydrated Human Amnion Chorion Membrane (dHACM) and PURION PLUS Processed Dehydrated Human Umbilical Cord (dHUC) Allografts.
- Bullard JD, Lei J, Lim JJ, Massee M, Fallon AM, Koob TJ. Evaluation of dehydrated human umbilical cord biological properties for wound care and soft tissue healing. J Biomed Mater Res B Appl Biomater. 2019;107(4):1035-1046.
- Dulemba J, Mirzakhani P, Istwan NB. Evaluation of Dehydrated Human Amnion/Chorion Membrane as an Adhesion Barrier in Women Undergoing Robotic Laparoscopy. Gynecol Obstet (Sunnyvale). 2016 Oct; 6:10.
- Angela F. Falany, MD, Obstetrics & Gynecology, Canton, GA.
- John A. Marascalco, MD, Dermatology, Grenada, MS.