
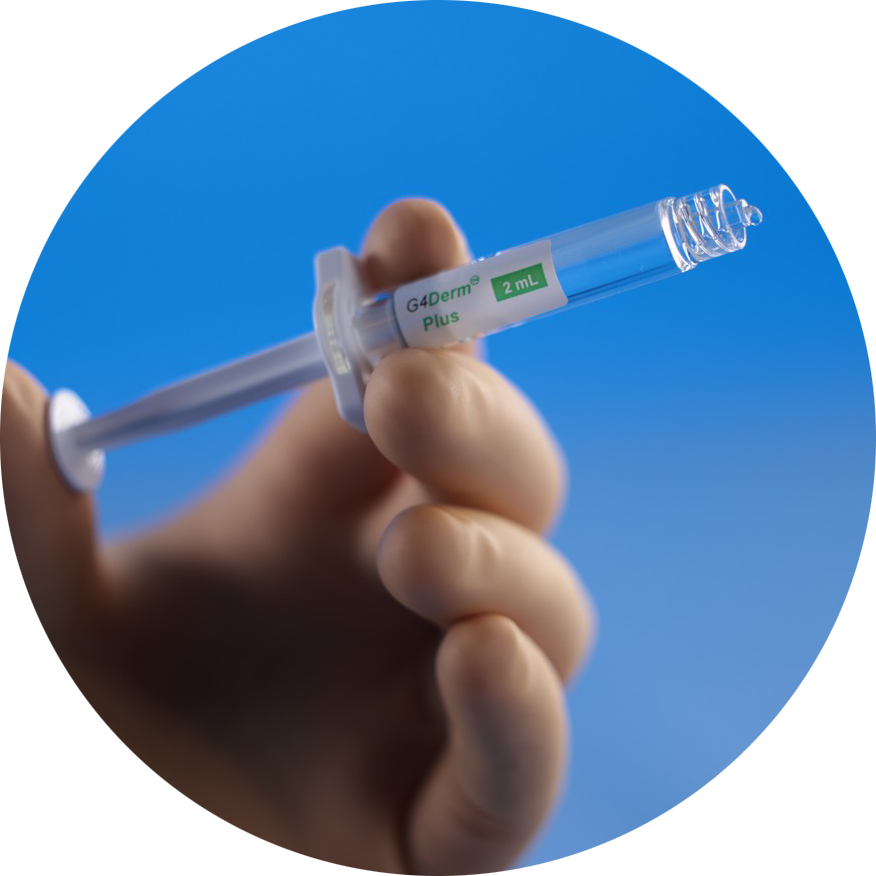
G4Derm® Plus is an acellular flowable Extracellular Matrix (ECM) designed to optimally manage complex wounds.
It supports healing and serves as an antibacterial barrier to protect the wound bed from external contamination.1 G4Derm Plus utilizes self-assembling peptide technology to form a scaffold that mimics the native ECM.1,2 The resorbable Biomimetic Matrix (BMM™) facilitates revascularization, granulation tissue formation, and re-epithelization.2,3
G4Derm Plus Mimics the Native ECM2,7
G4Derm Plus

Native ECM
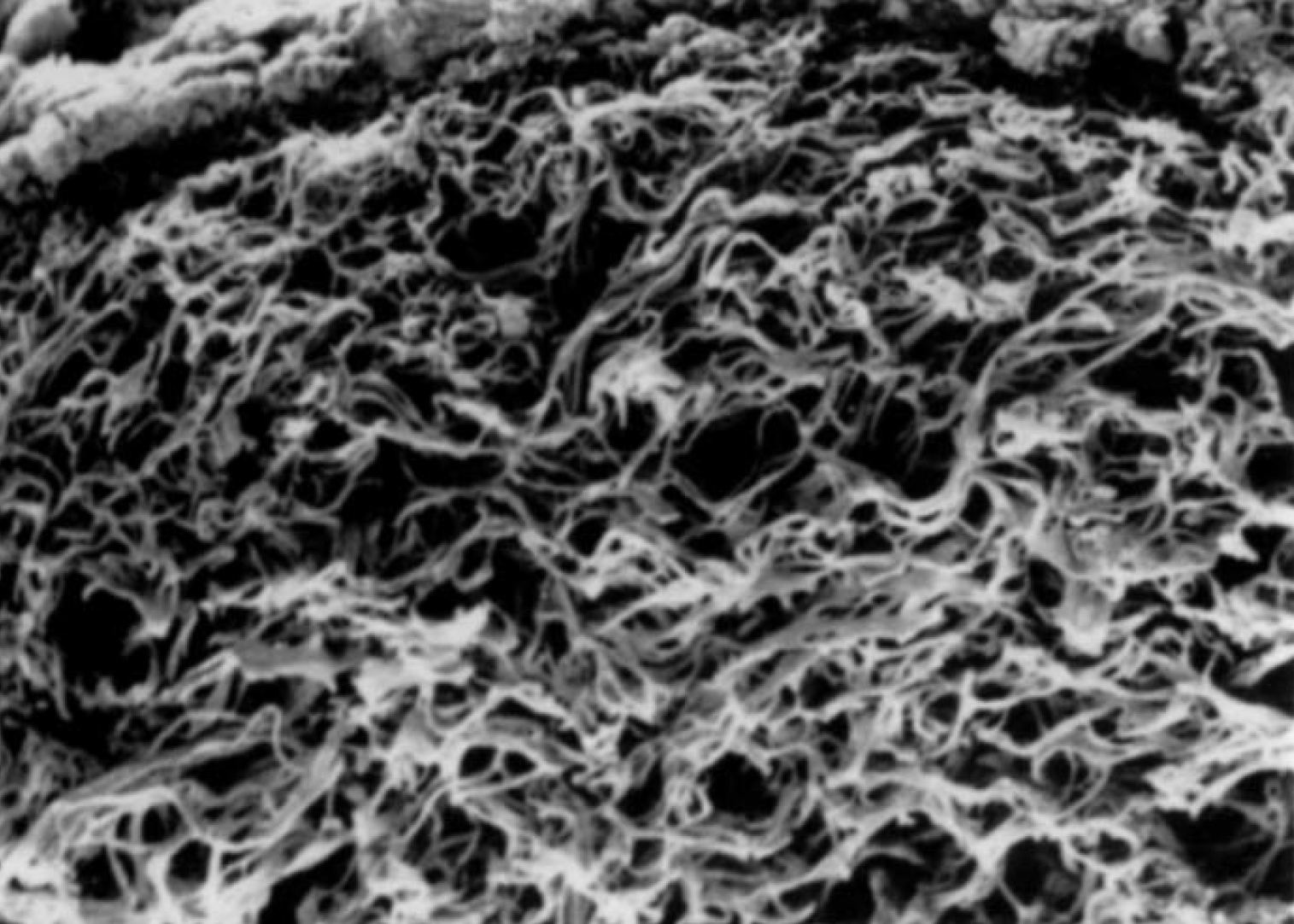
Scanning Electron Microscopy (SEM) magnification 5,000x and 270x, respectively.
Product Advantages
- Conforms to irregular wound geometries and eliminates dead space1,4
- Serves as a durable antibacterial barrier1,5
- Comes as a ready-to-use, pre-filled Syringe with a flexible Applicator Tip, and is shelf stable for up to 3 years1,6
G4Derm Plus is supplied in a box containing:
- One sterile single-use pre-filled Syringe
- One sterile single-use flexible Applicator Tip
- Instructions for Use and six patient labels
G4Derm Plus
| GTIN | Product # | Syringe Volume (mL) | Description | ~Wound Area Coverage (cm2) | ~ Coating Thickness (mm) |
| 00850058790138 | G4M200M | 2 mL | G4Derm Plus | 25 cm2 | 1 mm |
Indications for Use: G4Derm Plus may be used under the supervision of health care professionals for the local management of partial-and full-thickness wounds including pressure, and diabetic ulcers; lower extremity ulcers including those of venous, arterial and mixed etiology; surgical wounds; first-degree and partial thickness burns.
Healthcare professionals must use their own clinical judgment in evaluating appropriate treatment options for a particular patient. Treatment of a specific patient should be based on individual needs and the medical care deemed necessary by the patient’s treating physician and institutional protocols. Always refer to the package insert, product label, and/or instructions for use before using any MIMEDX product. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your MIMEDX representative if you have questions about MIMEDX products.
Resources

Product Brochure
Product Overview Presentation
MIMEDX is a registered trademark of MIMEDX Group, Inc. All other trademarks are owned by their respective owners.
- Data on File, FDA 510(k) submission package. Gel4Med, Inc.
- Data on File, Scanning Electron Microscopy of G4Derm Plus. Gel4Med, Inc.
- Data on File, G4Derm Plus Full-Thickness Excisional Wound Healing Study in Swine. Gel4Med, Inc.
- Data on File, Void-Filling Test of G4Derm Plus. Gel4Med, Inc.
- Data on File. Antibacterial Barrier Effectiveness of G4Derm Plus. Gel4Med, Inc.
- Data on File, G4Derm Plus Stability and Shelf-life. Gel4Med, Inc.
- Tsuji T, et al. J Microsc. 1979;115(2):165 173.








