Advanced Placental Allografts When Patients Are in Need
Burns are a major source of injury that, in the absence of care, can lead to lifelong functional loss and disfigurement. While split thickness skin autografts are the current standard of care for deep partial and full-thickness burns, this approach is associated with considerable complications. AMNIOBURN®, an advanced bilayer amnion/chorion membrane allograft, provides an alternative with strong clinical and scientific evidence for the treatment of burns.
Video Testimonial
Potential Areas of Use
- Points of Articulation
- Head/Face
- Hands
- Genitals
- Bone & Tendon
- Feet
AMNIOBURN Product Advantages
- Provides a protective barrier on wounds and thereby supports development of granulation tissue and the healing process
- Shelf-stable* with a 5-year shelf life
- PURION® process preserves Extracellular Matrix (ECM) components, including 300+ regulatory proteins1,2
- Compatible with Negative Pressure Wound Therapy (NPWT) and Hyperbaric Oxygen Therapy (HBOT)
*See Instructions for Use
Case Studies:
How Physicians Use Our Products
Case study 1
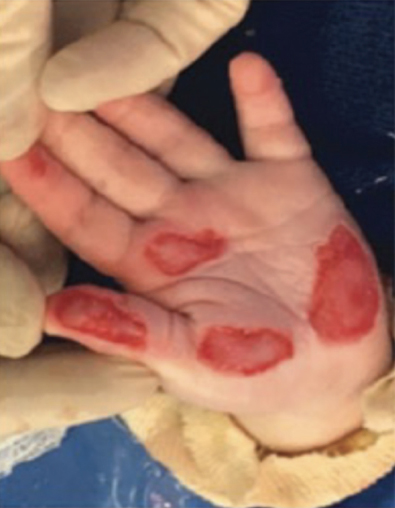
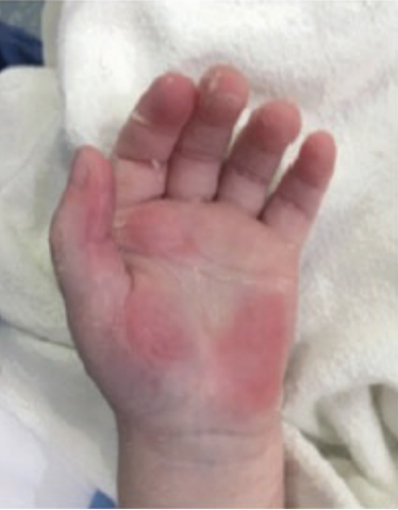
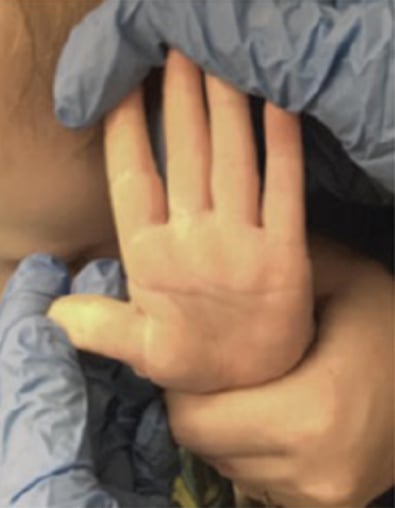
Full-Thickness Hand Burns to Fingers Bilaterally Treated With DHACM*3
 |
 |
 |
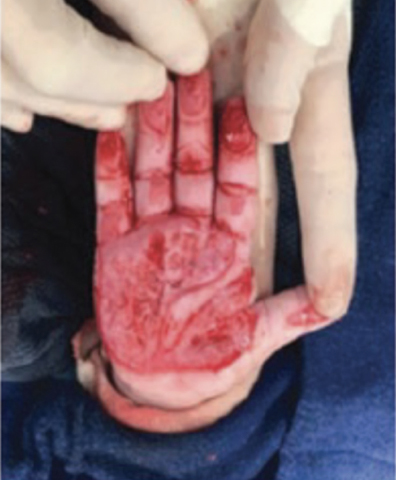 Initial debridement and application of DHACM (not pictured) Initial debridement and application of DHACM (not pictured) |
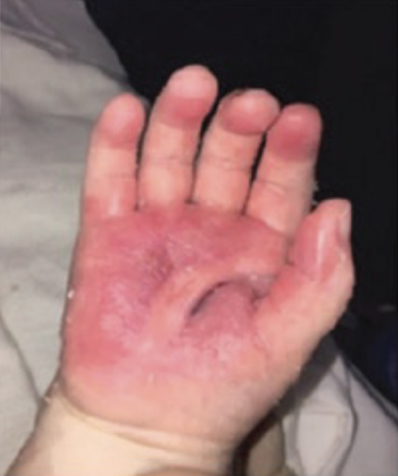 Postop Day 38: Complete closure Postop Day 38: Complete closure |
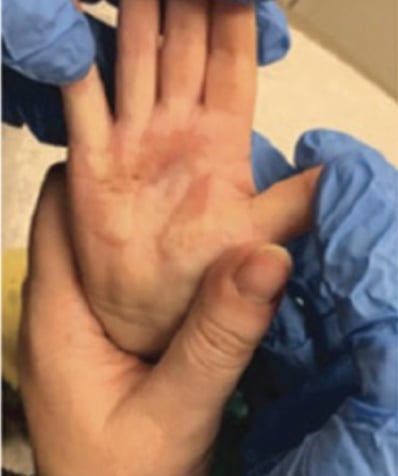 Postop Day 115: Observed no contractures Postop Day 115: Observed no contractures |
Case study 2
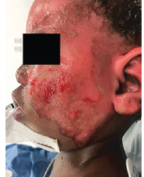
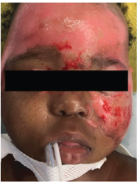
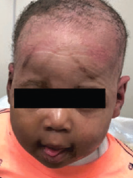
Superficial Partial-Thickness Facial Burn Treated with dHACM*3
|
Initial debridement (left lateral) |
Initial debridement (anterior) |
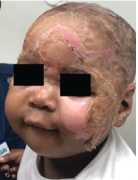 Postop Day 7 status-post DHACM application |
Complete closure |
Case study 3
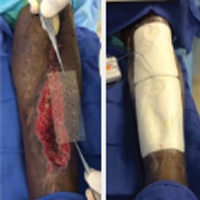
Full-Thickness Burn to Limb Treated With Fenestrated DHACM**+ NPWT4
Fenestrated DHACM + NPWT in comorbid burn patient with 10 cm x 15 cm laceration on LLE. Surgical debridement included tendon, soft tissue, and periosteum.
Comorbidities: Uncontrollable DM, smoking, CAD, PAD, COPD, HTN, asthma, and arthritis
Goal: STSG or Secondary Intention
 |
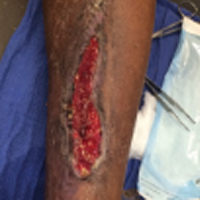 |
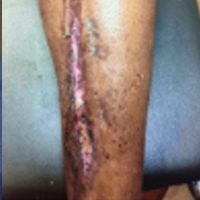 |
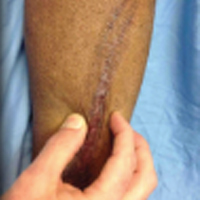 |
| Application of fenestrated DHACM + NPWT | Week 2 | Week 3: STSG avoided after only DHACM application | 5 months postop |
**This case used EPIXL FENESTRATED DHACM, which was discontinued in 2021.
Surgical Product Portfolio: Advanced Placental Allografts
MIMEDX offers a portfolio of advanced placental allografts in the surgical setting.
| AMNIOFIX® | AMNIOBURN® | AMNIOCORD® | AXIOFILL® | AMNIOEFFECT® | |
| Complex Soft-Tissue Deficit | ✓ | ✓ | ✓ | ✓ | ✓ |
| Large Area Coverage | ✓ | ✓ | ✓ | ✓ | |
| Affix Product with Suture* | ✓ | ✓ | |||
| Product Thickness Desired | ✓ | ✓ | ✓ | ||
| Exposed Bone, Tendon, or Hardware | ✓ | ✓ | ✓ | ✓ | |
| Reposition Product after Hydration | ✓ | ✓ | ✓ | ✓ | ✓ |
| Minimally Invasive Procedures | ✓ | ✓ | ✓ | ||
| Fenestrated Configuration to Allow Transfer of Exudate | ✓ |
References
- Moreno S, Massee M, Campbell S, Bara H, Koob TJ, Harper JR. PURION® processed human amnion chorion membrane allografts retain material and biological properties supportive of soft tissue repair. J Biomater Appl. 2024;39(1):24-39.
- MIMEDX Internal Report. MM-RD-00086, Proteome Characterization of PURION Processed Dehydrated Human Amnion Chorion Membrane (dHACM) and PURION PLUS Processed Dehydrated Human Umbilical Cord (dHUC) Allografts.
- Natasha Ahuja, Richard Jin, Colin Powers, Alexandria Billi, and Kathryn Bass, Pediatric Surgeons, Buffalo, NY.
- W. Dotie Jackson, MD, Plastic and Reconstructive Surgery, Jackson, MS.